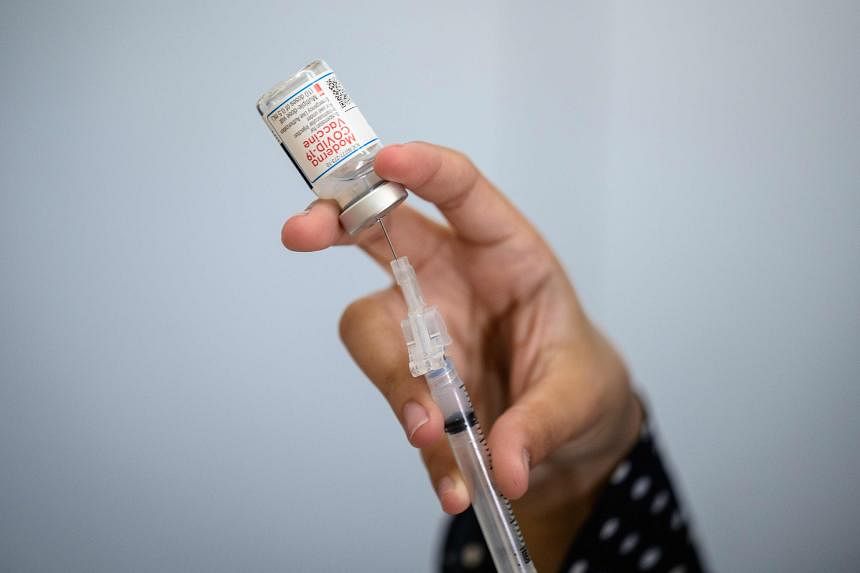
SILVER SPRING – The US Food and Drug Administration on Monday authorised updated Covid-19 vaccines that closely match the widely circulating Omicron variants, allowing the deployment of the shots to start this month.
The agency approved the shots, which target the XBB.1.5 subvariant, for emergency use from manufacturers Pfizer and its German partner BioNTech SE, and Moderna.
The FDA authorisation comes after a late summer rise in cases, at a time the new EG.5 subvariant of Omicron – nicknamed “Eris” – has begun to rapidly spread in the United States and in other parts of the world.
The US Centers for Disease Control and Prevention (CDC) advisers are due to meet on Sept 12 to recommend who should receive the shot. An endorsement by CDC director Mandy Cohen, expected shortly, should clear the way for the new shots. Cohen has said she expects the shots to roll out in September.
The Covid-19 public health emergency ended in May and the government has handed over the responsibility of vaccinations to the private sector. The demand for the vaccine has, however, dropped sharply since. REUTERS
Moderna ties up with Immatics to boost cancer vaccine development
US CDC says existing antibodies can work against new Covid-19 variant
Join ST’s Telegram channel and get the latest breaking news delivered to you.
p.st_telegram_boilerplate:before {
display: inline-block;
content: ” “;
border-radius: 6px;
height: 6px;
width: 6px;
background-color: #12239a;
margin-left: 0px;
margin-right: 13px;
}
a.st_boilerplate {
font-family: “SelaneWebSTForty”, Georgia, “Times New Roman”, Times, serif;
}